Reimbursement, Ordering, and Pricing
JUMP TO SECTION:
JUMP TO SECTION:
In light of the opioid crisis, Heron has engaged regulators and legislators to encourage changing payment incentives that lead to overprescribing of opioids. For Medicare patients, ZYNRELEF is reimbursed separately in both the hospital outpatient and ASC settings of care, billed using J0668.
For additional Medicare reimbursement details, see the table below.
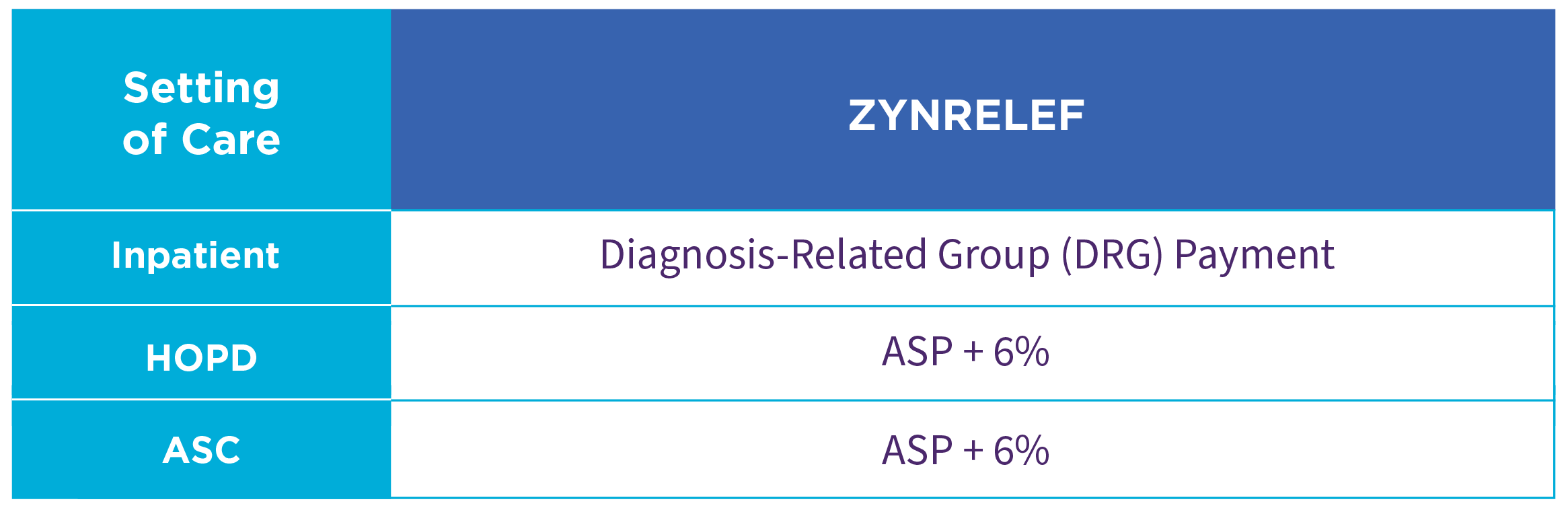
ASC: ambulatory surgical center.
ASP: average sales price.
CMS: Centers for Medicare & Medicaid Services.
HOPD: hospital outpatient department.

Separate payment for ZYNRELEF is available for many commercial patients. Commercial reimbursement and coding vary by payer and site of care; complete a benefit verification to confirm coverage and coding or request one by contacting Heron Connect®.
Download the ZYNRELEF Reimbursement and Billing Guide and the ZYNRELEF Reimbursement Overview for more details.
ZYNRELEF pricing supports broad access for patients and healthcare providers. ZYNRELEF is available through full-line wholesalers and specialty distributors; prime vendor discounts apply. GPO and sub-WAC pricing are also available.
ZYNRELEF is available in 2 volumes for convenience, minimum waste, and reduced average cost across the surgical mix.
| ZYNRELEF Pricing | ||
|---|---|---|
| Volume | Bupivacaine/Meloxicam | WAC |
| 14 mL | 400 mg/12 mg | $325.34 |
| 7 mL | 200 mg/6 mg | $162.67 |
GPO: group purchasing organization. WAC: wholesaler acquisition cost.
ZYNRELEF pricing as of January 1, 2026. Confirm current pricing with your Heron representative.
See Dosing and Administration to see which vial size is appropriate for example procedures.
Heron Connect offers customized support for ZYNRELEF billing and coding questions.
Reimbursement Counselors are available at
From answering questions about ZYNRELEF to providing updates on the reimbursement landscape, Heron representatives are here to help.
ZYNRELEF is indicated in adults for instillation to produce postsurgical analgesia for up to 72 hours after soft tissue and orthopedic procedures including foot and ankle, and other procedures in which direct exposure to articular cartilage is avoided.
Limitations of Use: Safety and efficacy have not been established in highly vascular surgeries, such as intrathoracic, large 4 or more level spinal, and head and neck procedures.
ZYNRELEF is contraindicated in patients with a known hypersensitivity (eg, anaphylactic reactions and serious skin reactions) to any amide local anesthetic, NSAIDs, or other components of ZYNRELEF; with history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs (severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients); undergoing obstetrical paracervical block anesthesia; or undergoing CABG.
Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient’s state of consciousness after application of ZYNRELEF. When using ZYNRELEF with other local anesthetics, overall local anesthetic exposure must be considered through 72 hours.
Hepatotoxicity: If abnormal liver tests persist or worsen, perform a clinical evaluation of the patient.
Hypertension: Patients taking some antihypertensive medication may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure.
Heart Failure and Edema: Avoid use of ZYNRELEF in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure.
Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of ZYNRELEF in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal failure.
Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs.
Risk of Joint Cartilage Necrosis and Degeneration with Unapproved Intra-articular Use: Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration.
Chondrolysis: Limit exposure to articular cartilage due to the potential risk of chondrolysis.
Methemoglobinemia: Cases have been reported with local anesthetic use.
Serious Skin Reactions: NSAIDs, including meloxicam, can cause serious skin adverse reactions. NSAIDs can also cause fixed drug eruption (FDE). FDE may present as a more severe variant known as generalized bullous fixed drug eruption (GBFDE), which can be life-threatening. If symptoms present, evaluate clinically.
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): If symptoms are present, evaluate clinically.
Fetal Toxicity: Due to the risk of oligohydramnios/fetal renal dysfunction and premature closure of the ductus arteriosus with NSAIDs, limit use of ZYNRELEF between about 20 to 30 weeks gestation, and avoid use after about 30 weeks.
Hematologic Toxicity: Monitor hemoglobin and hematocrit in patients with any signs or symptoms of anemia.
Drugs That Interfere with Hemostasis: Monitor patients for bleeding who are using ZYNRELEF with drugs that interfere with hemostasis (eg, warfarin, aspirin, SSRIs/SNRIs).
ACE Inhibitors, Angiotensin Receptor Blockers (ARBs), or Beta-Blockers: Use with ZYNRELEF may diminish the antihypertensive effect of these drugs. Monitor blood pressure.
ACE Inhibitors and ARBs: Use with ZYNRELEF in elderly, volume-depleted, or those with renal impairment may result in deterioration of renal function. In such high-risk patients, monitor for signs of worsening renal function.
Diuretics: NSAIDs can reduce natriuretic effect of furosemide and thiazide diuretics. Monitor patients to assure diuretic efficacy including antihypertensive effects.
Infertility: NSAIDs are associated with reversible infertility. Consider avoidance of ZYNRELEF in women who have difficulties conceiving.
Severe Hepatic Impairment: Only use if benefits are expected to outweigh risks; monitor for signs of worsening liver function.
Severe Renal Impairment: Not recommended.
Most common adverse reactions (incidence ≥5%) in controlled clinical trials with ZYNRELEF are soft tissue procedures: vomiting and orthopedic procedures: constipation and headache.
Report side effects to Heron at 1-844-437-6611 or to FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
ZYNRELEF is indicated in adults for instillation to produce postsurgical analgesia for up to 72 hours after soft tissue and orthopedic procedures including foot and ankle, and other procedures in which direct exposure to articular cartilage is avoided.
Limitations of Use: Safety and efficacy have not been established in highly vascular surgeries, such as intrathoracic, large 4 or more level spinal, and head and neck procedures.
Please see full Prescribing Information, including Boxed Warning and updated Warnings and Precautions for serious skin reactions caused by nonsteroidal anti-inflammatory drugs (NSAIDs).
How could you incorporate ZYNRELEF as the foundation of your institution’s postoperative pain management strategy? Connect with us to find out.
Fields marked with an asterisk (*) are required.
In the meantime, you can learn more about ZYNRELEF by viewing our Frequently Asked Questions or downloading our Resources.
844-HERON11 (844-437-6611)